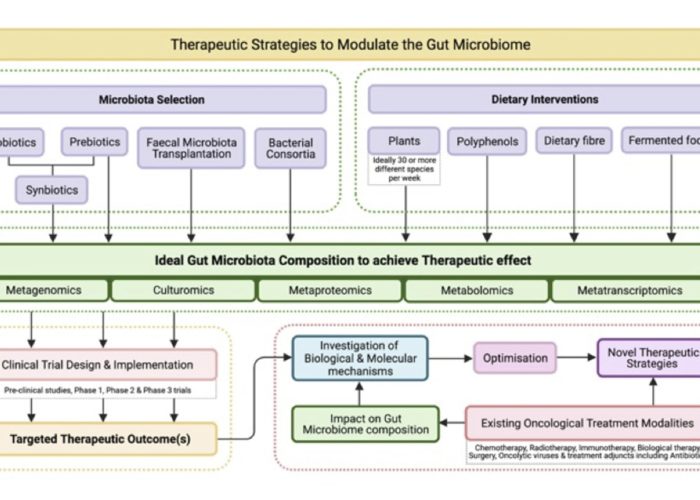
Category: Microbiome
Meet the Microbiome & Cancer Working Group

What can we do differently today to improve outcomes for cancer patients?
One clear, evidence-based takeaway from today’s keynote at the Society for Immunotherapy of Cancer (SITC): clinicians should be judicious when prescribing antibiotics before or during immunotherapy. Multiple studies have shown that antibiotic use in these settings is associated with significantly shorter overall survival.
In her keynote address, Dr. Jen Wargo of MD Anderson Cancer Center also announced the launch of the Microbiome and Cancer Working Group — a new international initiative to deliver clear, evidence-based recommendations on how the microbiome can improve outcomes for every cancer patient.
Co-chaired by Dr. Arielle Elkrief of CHUM and Dr. Wargo, this global, multi-disciplinary team will bring together leading experts across oncology, immunology, microbiology, and public health to:
- Synthesize current scientific understanding of the microbiome and cancer
- Identify key research gaps
- Establish standards for evidence generation
- Define a clear path for integrating microbiome science into clinical care
Seerave is proud to sponsor this landmark initiative and to help connect the brightest minds working at the intersection of cancer and the microbiome. We believe that microbiome science has the power to transform cancer outcomes — not in decades, but starting today.
Further Reading: Gut Microbiota in Immuno-Oncology: A Practical Guide for Medical Oncologists With a Focus on Antibiotics Stewardship
Continue Reading
Related Articles
Seerave invests in Florey Biosciences and takes aim at mitigating impacts of antibiotic use

Key Points
- Seerave Ventures has made a pre-seed investment in Florey Biosciences, a spinout of the Wyss Institute at Harvard University
- Florey is developing an engineered yeast strain designed for the management of antibiotic damage to the gut microbiome
- As a founding partner, Seerave will contribute strategic and operational support alongside its capital investment
October 1, 2024 – Today, we are announcing that Seerave Ventures, the investment arm of the Foundation, has made a new investment in Florey Biosciences, a synthetic biology company with a novel technology platform that can deliver any protein of interest to the gut. Florey’s lead program is a medical food intended for the dietary management of antibiotic damage to the gut microbiome.
Florey’s technology is a promising innovation for addressing the impacts of antibiotic use. It speaks to the strategic commitment that we recently announced to mitigate the impacts of microbiome modifiers on cancer outcomes and deliver meaningful benefits to patients within five years.
Immunotherapy is one of the most promising new frontiers of cancer treatment, but overall survival is about half as long for patients who received antibiotics before immunotherapy. Often, taking antibiotics is an unavoidable and life-saving measure. It can, however, compromise the microbes that live in our guts and that play a profound role in how our immune systems function.
“We see our support of Florey as a key part of our efforts to meaningfully improve patients’ lives within the next five years,” said Manuel Fankhauser, CEO of the Seerave Foundation, in a press release issued by Florey on Oct 1.
As part of Seerave’s involvement, Mark Smith, Venture Lead at Seerave, has joined the board of Florey Biosciences.
For more information:
Continue Reading
Related Articles
Seerave Announces Strategic Focus on Antibiotics and Cancer Immunotherapy
Over the past twelve months, the Seerave Foundation has convened large parts of its network to identify how to best position its science and venture philanthropy platform to deliver meaningful improvements to cancer care within the next five years. Drawing from these conversations, which have been partially summarized in a recent Cancer Cell perspective, we would like to announce a new strategic focus for the Foundation: mitigating the impacts of antibiotics and other microbiome modifiers on cancer patients’ outcomes.
An estimated 30% of patients take antibiotics prior to immunotherapies like immune checkpoint inhibitors, and analyses of more than 40,000 patients have found a strong association between antibiotic use, immune system function, and poorer responses to these promising new treatments. Immunotherapies still only work in a fraction of cancer patients, and it has become a major effort of oncology researchers to uncover why.
Members of Seerave’s research network put forth some of the first studies and meta analyses looking closely at the relationship between antibiotic use, immunotherapy, and poorer patient outcomes, and the follow-up investigations on mechanism and causality.
Building on this work, Seerave’s strategic focus is designed to help the Foundation measurably improve cancer patients’ lives within five years. Seerave aims to clarify how antibiotics and other microbiome modifiers impact patient outcomes, inform practical advice to clinicians and patients, and propel the development of new tools and technologies.
In pursuit of these goals, we are privileged to welcome our first Scientific Advisors, Martin Blaser, MD and Per Falk, MD, PhD to advance the Foundation’s research agenda and bring scientific excellence to its programs.
- Dr. Martin Blaser holds the Henry Rutgers Chair of the Human Microbiome at Rutgers University, and serves as Director of the Center for Advanced Biotechnology and Medicine. A physician and microbiologist, Dr. Blaser has driven seminal research on the relationship of the human microbiome with health and diseases, including asthma, obesity, diabetes, and cancer. Dr. Blaser has served as President of the Infectious Diseases Society of America, Chair of the Board of Scientific Counselors of the National Cancer Institute, Chair of the Advisory Board for Clinical Research of the NIH, and Chair of the Presidential Advisory Council for Combatting Antibiotic Resistant Bacteria (PACCARB). He has authored over 600 original scientific articles, holds 24 U.S. patents, and is the author of Missing Microbes.
- Dr. Per Falk has been investigating and advancing microbiome science for over 30 years, most recently as President and Chief Scientific Officer of Ferring Pharmaceuticals, where he oversaw the development of one of the first microbiome therapies to receive FDA approval. Prior to joining Ferring, Dr. Falk held executive and senior leadership positions at Novo Nordisk and AstraZeneca in research, medical and clinical development roles. He has held the role of Associate Professor at the Karolinska Institute, Sweden and the Washington University School of Medicine, USA.
Seerave is also delighted to welcome two new team members, Mark Smith, PhD and Carolyn Edelstein, to lead some of its new activities. Our approach will be organized under the following strategic pillars:
- Data Integration: Build a data consortium that collaboratively integrates and interrogates clinical data on microbiome modifiers in cancer immunotherapy from leading hospitals and research institutions. Led by Laura Mählmann
- Policy and Practice: Empower oncologists and other key stakeholders with practical, evidence-based advice that reflect the latest understanding of the role of microbiome modifiers in cancer care. Led by Carolyn Edelstein
- Novel Tools: Foster the development of new tools and services for oncologists through Seerave Ventures, our hands-on impact investment vehicle that supports a select number of mission-aligned companies with the potential to transform patient care. Seerave Ventures will be led by Mark Smith
- Community and Collaboration: Cultivate community across disciplinary silos and geography by creating and empowering a self-sustained, mission-aligned network of relevant stakeholders, including clinical researchers, clinical practitioners, public health experts, entrepreneurs, and patient advocates. Led by Laura Mählmann
A legacy of bringing new insights to cancer care
Almost a decade ago, after seeing how an emphasis on microbiome health brought new hope during his wife’s cancer journey, David Rees established the Seerave Foundation with a vision of bringing precision medicine and groundbreaking scientific insights on the microbiome and immune system function to cancer care. At the time, an appreciation of the microbiome’s relevance to cancer was far from mainstream. Today, it is seen as one of the most promising new arenas in the search for therapeutic breakthroughs. Seerave’s research network has driven many of the insights that have mobilized the field, including:
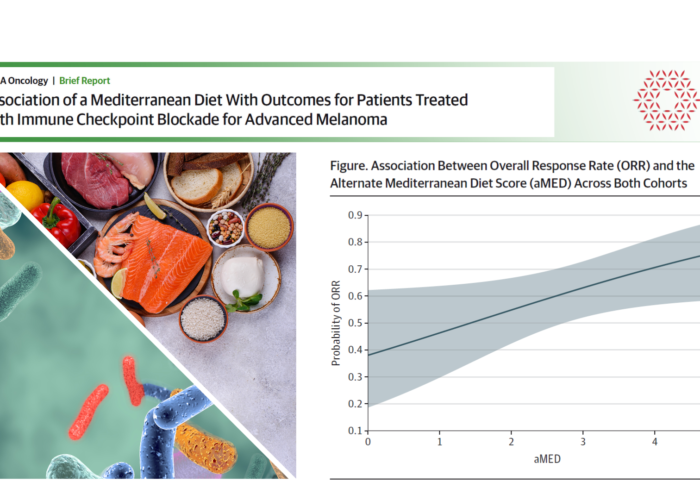
- The Mediterranean diet is associated with improved outcomes in melanoma patients following immune-checkpoint blockade [1]
- The microbiome is different in responders vs. non-responders to immune-checkpoint blockade [2] [3], there are plausible mechanisms by which microbiota may influence therapeutic response, and biomarkers of cancer-associated microbiome dysbiosis [4]
- Fecal microbiota transplantation may improve immune-checkpoint-blockade outcomes [5]
Beyond its role in catalyzing research, Seerave has supported the translation of these and other key discoveries into a wide array of products, from new public health tools and biotechnology companies to public interest projects like microbiome conservation initiatives and critically acclaimed documentaries.
We look forward to sharing updates of our progress in the coming months. We would also keenly welcome feedback and opportunities to collaborate, and we would love to hear if our mission and work resonates with you and is as close to your heart as it is to ours.
Continue Reading
Related Articles
Longitudinal PRIMM Data published in Nature Medicine
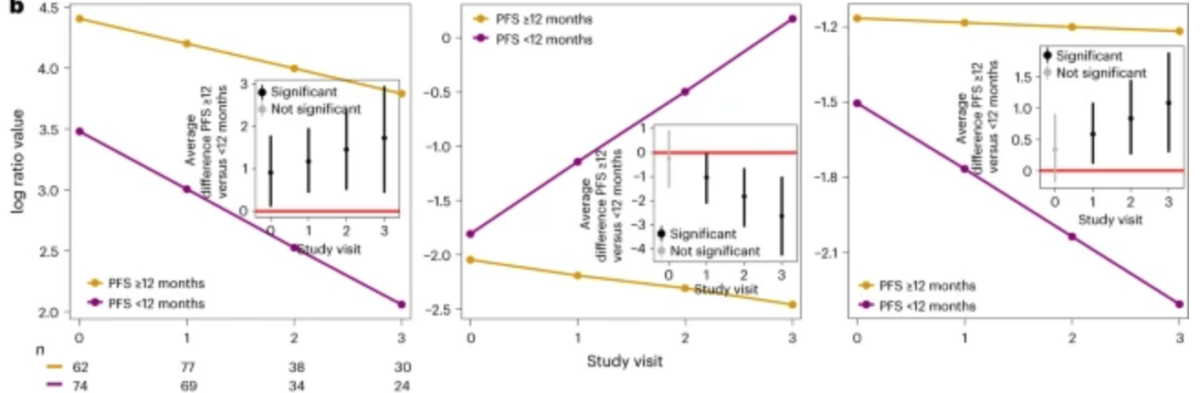
The first longitudinal assessment of metagenomic signatures along the ICI/cancer treatment journey has been published by our grantees in Nature Medicine. The publication entitled “Longitudinal gut microbiome changes in immune checkpoint blockade in advanced melanoma” expands our understanding on how the microbiome may impact response to cancer treatment.
Notably, the study introduces “balance scores” – a collection of bacteria that play crucial roles in both positive and negative associations with treatment response, not just at the beginning but throughout the treatment process.
🔍 So, why is this important?
- It highlights the necessity of longitudinal sampling, suggesting that a single baseline gut microbiome sample might not capture how bacteria influence the immune system and treatment response over time.
- The published balance score, comprising approximately 9 key bacteria, holds potential for translation into diagnostic tools and therapeutic interventions.
- As a next step the team is analyzing multi-omic data, especially adding a metabolomic layer, promising to further enhance our ability to predict treatment outcomes by understanding microbial function.
Huge congratulations to this impactful collaborations among Johannes Björk, Laura Bolte, Geke Hospers, Rinse Weersma from UMCG; Karla Lee, Veronique Bataille and Tim Spector from KCL and Lisa Derosa, Laurence Zitvogel (IGR), Nicola Segata (University of Turin) and many more.
Please find the full open-access publication here: https://www.nature.com/articles/s41591-024-02803-3
This publication adds to a series of publications from the same cohort, find them here:
Overall, this is paving the way for more personalized and effective cancer treatments.
Continue Reading
Related Articles
More effective cancer immunotherapy thanks to the gut microbiome
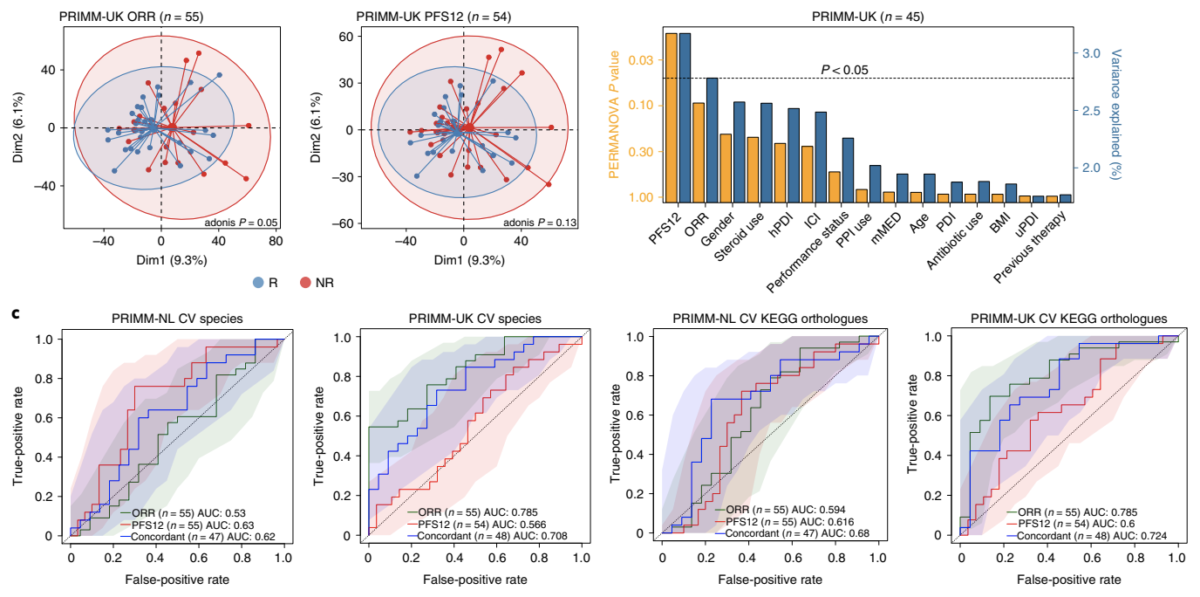
For more than four years, Seerave Foundation has been supporting the outstanding research teams at King’s College London (UK), UMCG (NL) and University of Trento (I) to find out whether there is a link between the presence and function of the gut microbiome and the outcome of cancer immunotherapy. We are thrilled to see this milestone work being published in Nature Medicine!
Congrats to all the authors and especially Karla Lee, Andrew Thomas, Laura Bolte, Johannes Björk, Laurence Zitvogel, Veronique Bataille, Geke Hospers, Tim Spector, Rinse Weersma and Nicola Segata from the Seerave network!
In summary, the team found that the gut microbiome has a relevant, but cohort-dependent, association with the response to ICIs. A panel of species, including Bifidobacterium pseudocatenulatum, Roseburia spp. and Akkermansia muciniphila, associated with responders was identified, but no single species could be regarded as a fully consistent biomarker across studies. Overall, the role of the human gut microbiome in ICI response appears more complex than previously thought, extending beyond differing microbial species simply present or absent in responders and non-responders.
We hope that this open access publication will drive more research into finding the mechanistic answers needed to drive personalised microbiome-targeted interventions forward. Dedicated papers regarding the association of different diet patterns with response in the same cohorts will be published separately in the coming months (as well as papers including proteomic, glycomic and metabolomic analysis).
Stay tuned!
Source: https://www.nature.com/articles/s41591-022-01695-5.pdf
Continue Reading
Related Articles
The Microbiota Vault is being launched in Switzerland

In 2018, Seerave embarked on the vision of Martin Blaser and Gloria-Maria Dominguez-Bello aiming to establish “The Microbiota Vault” – an initiative examining the potential of setting up a global repository of human microbiota. This repository, a “Noah’s Ark for microbes” would focus on the preservation of the current global diversity of the human microbiota, on backing-up global research collections and to further explore the role of the microbiome in health and disease. The launch of this important project was only possible thanks to philanthropic co-impact including Gebert Rüf Foundation and Rockefeller Foundation joining forces.
We hope others will join in as well!
For our German-speaking network: please have a look at the recent NZZ article for more in depth reporting.
Continue Reading
Related Articles
The gut microbiome: what the oncologist ought to know
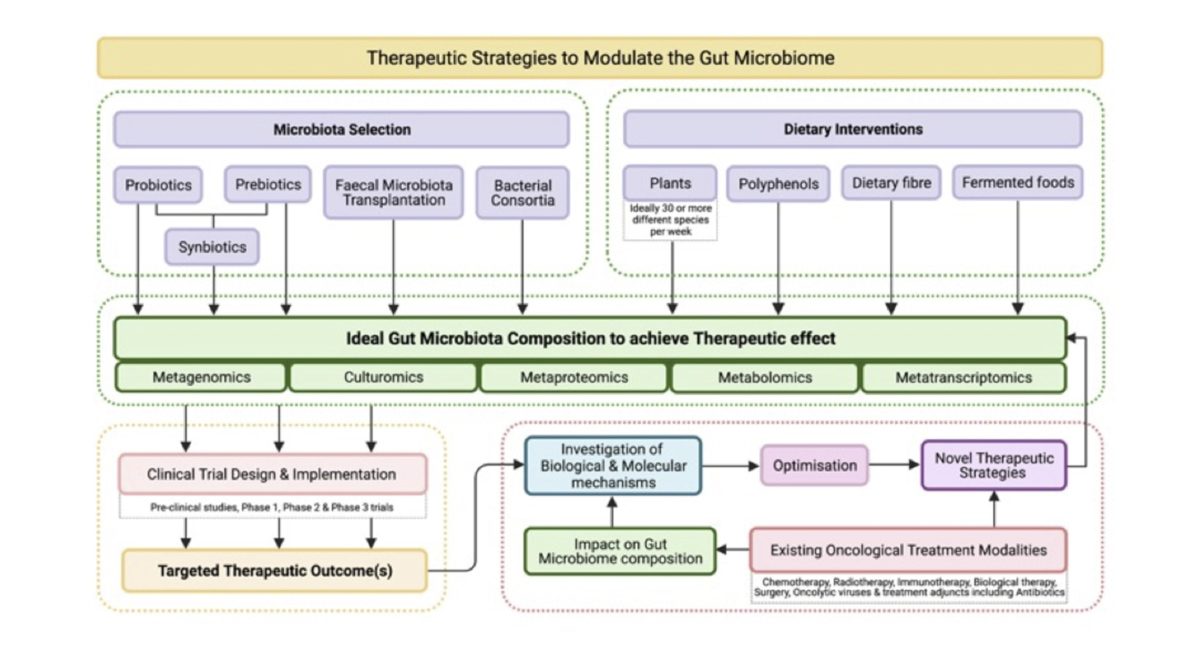
The gut microbiome (GM) has been implicated in a vast number of human pathologies and has become a focus of oncology research over the past 5 years. The normal gut microbiota imparts specific function in host nutrient metabolism, xenobiotic and drug metabolism, maintenance of structural integrity of the gut mucosal barrier, immunomodulation and protection against pathogens.
Strong evidence is emerging to support the effects of the GM on the development of some malignancies but also on responses to cancer therapies, most notably, immune checkpoint inhibition.
Tools for manipulating the GM including dietary modification, probiotics and faecal microbiota transfer (FMT) are in development. Current understandings of the many complex interrelationships between the GM, cancer, the immune system, nutrition and medication are ultimately based on a combination of short‐term clinical trials and observational studies, paired with an ever-evolving understanding of cancer biology.
The next generation of personalised cancer therapies focusses on molecular and phenotypic heterogeneity, tumour evolution and immune status; it is distinctly possible that the GM will become an increasingly central focus amongst them.
The aim of this review is to provide clinicians with an overview of microbiome science and our current understanding of the role the GM plays in cancer.
Continue Reading
Related Articles
Happy #WorldMicrobiomeDay 2021! 🎉

On World Microbiome Day we celebrate all things microbial, all around the world.
Today we would like to raise special attention to one of our initiatives: The collaboration between MyFoodRepo @EPFL and Microsetta @UCSD. Together they aim to roll out a US-wide citizen science project aiming to collect as much information as possible on everyday food items we consume. This information will help to train an algorithm that enables researchers in the future to better track nutrition and give more profound insights. Therefore…
We ask all of you to contribute to the microbe revolution: the “FoodRepo Scan” app is a new way #CitizenScientists can help advance the understanding between diet & microbes! Download the app on iOS or Android and start adding barcodes and images of food items you have at home or encounter in any store. The data will then become publicly available on FoodRepo. This will create a hugely valuable dataset, available to anyone in the world, with which research into nutrition and health will be accelerated. For example, it will serve as a nutritional library for the MyFoodRepo platform, a new AI-assisted tool that allows tracking of high dimensional nutritional data during clinical studies. Become part of tomorrow’s science today!
The collaboration between Microsetta and the Digital Epidemiology team at EPFL is made possible thanks to the support from the Seerave Foundation for nutrition and microbiome research @Seerave Foundation; please see http://www.seerave.org/projects for more details.
Continue Reading
Related Articles
Long-term dietary patterns are associated with pro- inflammatory and anti-inflammatory features of the gut microbiome

Researchers found that specific foods and nutrients correlated with species known to have mucosal protection and anti-inflammatory effects, according to data published in Gut.
“We identified dietary patterns that consistently correlate with groups of bacteria with shared functional roles in both health and disease,” Laura A. Bolte, BSc, from the department of gastroenterology and hepatology, University of Groningen and University Medical Centre Groningen, Groningen, the Netherlands, and colleagues wrote. “Moreover, specific foods and nutrients were associated with species known to infer mucosal protection and anti-inflammatory effects. We propose microbial mechanisms through which the diet affects inflammatory responses in the gut as a rationale for future intervention studies.”
Bolte and colleagues analyzed the relationship between 173 dietary factors and the microbiome of 1,425 patients from different cohorts including Crohn’s disease, ulcerative colitis, irritable bowel syndrome and the general population. They profiled gut microbial composition and function with shotgun metagenomic sequencing assessed dietary intake via food frequency questionnaires.
“We performed unsupervised clustering to identify dietary patterns and microbial clusters,” Bolte and colleagues wrote. “Associations between diet and microbial features were explored per cohort, followed by a meta-analysis and heterogeneity estimation.”
The researchers identified 38 links between dietary patterns and microbial clusters. In their meta-analysis of healthy individuals and patients with IBS, Crohn’s disease and UC, investigators found 61 individual foods and nutrients correlated with 61 species and 249 metabolic pathways.
“Processed foods and animal-derived foods were consistently associated with higher abundances of Firmicutes, Ruminococcus species of the Blautia genus and endotoxin synthesis pathways,” Bolte and colleagues wrote. “The opposite was found for plant foods and fish, which were positively associated with short-chain fatty acid-producing commensals and pathways of nutrient metabolism.”
DOI: doi:10.1136/gutjnl-2020-322670.
Story Source: https://www.healio.com/news/gastroenterology/20210405/diet-patterns-linked-with-gut-proinflammatory-antiinflammatory-features
Continue Reading
Related Articles
Ketogenic diet and ketone bodies enhance the anticancer effects of PD-1 blockade
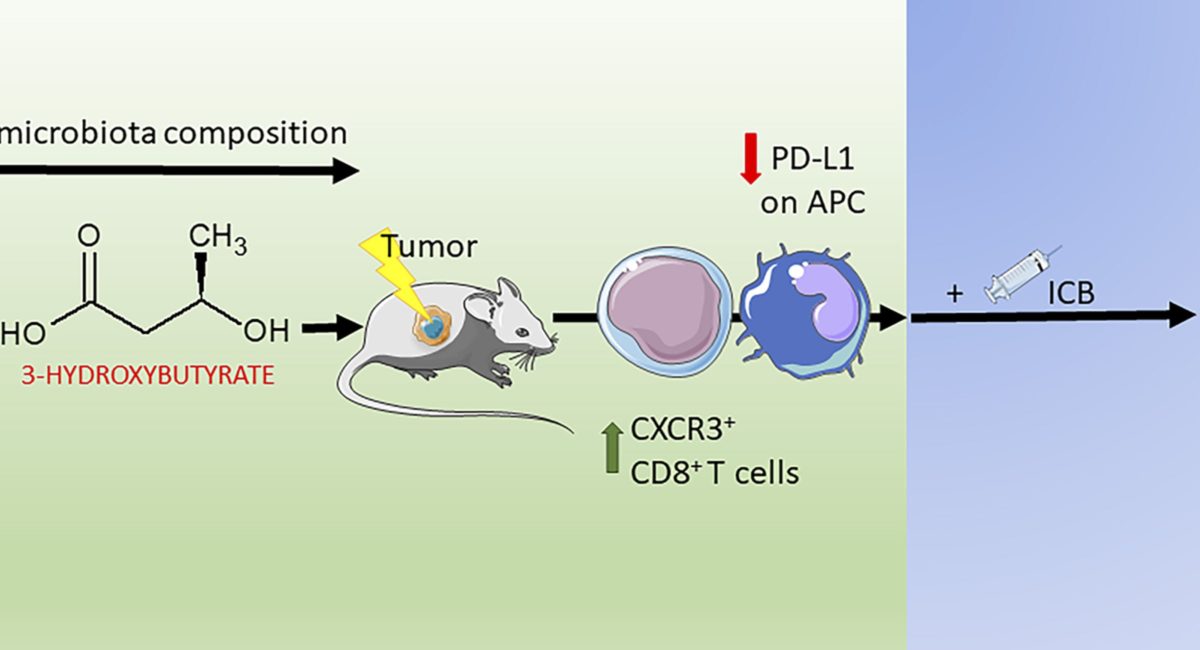
Limited experimental evidence bridges nutrition and cancer immunosurveillance. Here, the team at IGR Paris shows that ketogenic diet (KD) or its principal ketone body, 3-hydroxybutyrate (3HB), most specifically in an intermittent scheduling, induced T cell-dependent tumour growth retardation of aggressive tumour models.
In conditions in which anti-PD-1, alone or in combination with anti-CTLA-4, failed to reduce tumour growth in mice receiving a standard diet, KD or oral supplementation of 3HB reestablished therapeutic responses. Supplementation of KD with sucrose (which breaks ketogenesis, abolishing 3HB production) or with a pharmacological antagonist of the 3HB receptor GPR109A abolished the anti-tumour effects. Mechanistically, 3HB prevented the ICB-linked up-regulation of PD-L1 on myeloid cells while favouring the expansion of CXCR3+ T cells. KD induced compositional changes of the gut microbiota with distinct species such as Eisenbergiella massiliensis commonly emerging in mice and humans subjected to carbohydrate low diet interventions and highly correlating with serum concentrations of 3HB. Altogether, these results demonstrate that KD induces a 3HB-mediated antineoplastic effect that relies on T-cell mediated cancer immunosurveillance.
Continue Reading